The true facts and science being probiotics supplements.
Probiotic supplements are not fully understood and there is a lot of misunderstanding surrounding this category of products. This paper provides the facts and science supporting probiotic supplements.
Introduction
This technical paper provides professionals with succinct and technically accurate answers to the most regularly asked questions on probiotic bacteria and probiotic products.
- What are the health benefts of probiotic bacteria?
- How do probiotic bacteria confer health benefts?
- What is the correct potency for probiotic products?
- Why is probiotic stability important? Should probiotics be refrigerated?
- Are probiotic bacteria safe?
- What determines the effectiveness of probiotic bacteria?
- Are probiotic bacteria recommended and safe for children?
- Why have probiotic health claims been rejected by the EU?
- How are probiotic bacteria manufactured?
What are the health benefits of probiotics?
The scientific trials and data supporting the health benefits of probiotics is growing on a weekly basis. In fact, there is no other ingredient or product available in pharmacies or health food stores which can provide customers with such a wide-ranging list of health benefits, supported by science.
The health benefits of probiotics can be divided into two categories:
- Well-established health benefits for which there is su cient scientific support. See Table 1.
- Emerging & exciting new health benefits, which have some scientific support but which require further study. See Table 2.
Table 1: Established health benefits of probiotic supplementation
Antibiotic-induced diarrhoea 1,2
Antibiotics kill “friendly” bacteria together with pathogenic microorganisms. This disruption of the gut microflora causes diarrhoea. Probiotic bacteria restore the positive balance of “friendly” bacteria in the intestine and treat antibiotic-induced diarrhoea.
Traveller’s diarrhoea 3
Traveller’s diarrhoea is a common problem occurring in up to 50% of travellers and is caused by exposure to pathogenic microorganisms in new environments, food and water. Probiotic bacteria are highly effective in inhibiting these pathogens and prevent traveller‘s diarrhoea.
Gastroenteritis (stomach bugs) 4,5,6
Probiotic bacteria have been established as beneficial in preventing and treating the symptoms of gastroenteritis (stomach bugs), particularly acute watery diarrhoea.
Irritable bowel syndrome (IBS) 7,8,9
Irritable bowel syndrome (IBS) is a condition experienced by 20-15% of adults, often linked to stress. IBS causes altered bowel habits and abdominal discomfort or pain. Probiotics are highly e ective in reducing the symptoms of IBS.
Urinary tract and vaginal infections 11,12
Oral consumption of probiotics can positively affect the balance of probiotic bacteria in the vagina. 10 Probiotic bacteria provide a barrier to the pathogenic bacteria that cause urinary tract infections and prevent the growth of bacteria/yeasts causing vaginal infections e.g. candidiasis and bacterial vaginosis.
Pregnancy 17,38
There is very strong scientific evidence that prenatal supplementation with probiotic bacteria results in a significant reduction in atopic conditions in the infant, particularly atopic eczema.
Eczema (atopic dermatitis) 18,19
Clinical trials have shown that probiotic bacteria have a modest role in the treatment of atopic eczema, especially in children. The benefits of probiotics were clearer in individuals with moderately severe rather than mild eczema.
Helicobacter pylori infection 20,21
Certain lactobacilli probiotic bacteria have been shown to inhibit or kill H.pylori.
Immune stimulation 32,33
Preliminary studies show that probiotic bacteria can stimulate and regulate the immune response in the body and may contribute in reducing the risk or duration of certain infections.
Lactose intolerance 39,40
Lactobacilli probiotic bacteria convert undigested lactose into lactic acid in the intestine, thereby reducing the discomfort caused by lactose intolerance. Lactobacilli probiotic bacteria can be combined with lactase enzyme for greater synergistic effect.
Oral health and dental caries 13,14,15,16
Probiotics produce anti-bacterial compounds that have a positive effect on oral health, including plaque formation, bad breath, dental caries and gum bleeding or inflammation (gingivitis).
Table 2: Emerging health benefits of probiotic supplementation
Allergy prevention 34
There have been trials and research demonstrating the ability of probiotic bacteria in reducing allergic responses in humans (especially children), for example from food or pollen.
Cholesterol lowering 22,23
There is some evidence that probiotic bacteria may lower total serum cholesterol and LDL “bad” cholesterol.
Functional constipation 24
The role of probiotic bacteria in treating functional constipation is still at an investigational stage, but there are some clinical studies that suggest probiotics may help.
Respiratory tract infections 25
There are clinical trials supporting the theory that probiotic supplementation can reduce the severity and duration of respiratory tract infections.
Infant health and growth 26,27
Clinical studies are ongoing on the benefits of probiotic supplementation in infant formulas. Research has shown that probiotic bacteria supplementation can increase infant height and weight in the first 6 months of life.
Infant colic 28
One clinical study found a 65% reduction in infantile colic after 28 days in infants being given probiotic bacteria. More research is required to confirm these initial findings.
Irritable bowel disorders
There is some limited scientific evidence that suggest probiotic bacteria may help alleviate the symptoms of Crohn’s disease and ulcerative colitis through their anti-inflammatory effect.
Autism 35
Exciting and emerging research is ongoing.
Diabetes 36,37
Exciting and emerging research is ongoing.
How do probiotics confer health benefits?
The human intestinal microflora (see glossary) constitutes a complex ecosystem that plays a vital role in human health and disease, beyond gut and digestive health. There are numerous mechanisms of interaction between the cells in our body and probiotic bacteria, which have a direct impact on our health and well-being. In fact, there are 10 times more microorganisms in our gut, than we have cells in our body.
The modes of action of probiotic bacteria is an exciting and developing area of scientific research. Currently, not all the modes of action have been fully researched or completely understood.
Inhibition of pathogenic microorganisms
The ability of probiotic bacteria to stop pathogenic microorganisms (see glossary) from establishing in the gut is called “colonisation resistance”. Probiotic bacteria prevent the colonisation of pathogenic microorganisms in the gastrointestinal system (see glossary), keeping them at such low population levels, that they are unable to express their pathogenicity or
detrimentally affect the health of the individual.
Probiotic bacteria are highly resilient and compete with pathogenic microorganisms for adhesion sites and nutritional sources in the gastrointestinal system. Some probiotic bacteria also produce inhibitory compounds (see glossary) that further prevent the colonisation of pathogenic microorganisms. These compounds include lactic acid, hydrogen peroxide
and also anti-bacterial compounds, known as bacteriocins.
Protective intestinal barrier
Probiotic bacteria reinforce the intestinal lining by adhering to the cells in the intestine wall. As a result probiotic bacteria can positively influence absorption and prevent potentially harmful substances from crossing into the blood stream.
Stimulating the immune system
Probiotic bacteria play a positive role in regulating and activating immune function, improving the state of readiness of the immune system and alleviating the symptoms of immune-based conditions such as allergies and eczema.
The gastrointestinal system contains a large number of immune cells and compounds, and therefore plays an important role in the activation and effectiveness of the body’s whole immune system. The immune cells and compounds in the gastrointestinal system are in close proximity to probiotic bacteria, which are thought to influence optimal immune function throughout the body.
Detoxitication and metabolism
Metabolism (see glossary) refers to the chemical reactions that occur inside the cells of probiotic bacteria and which uses substances found in the probiotic bacteria surroundings and environment.
Probiotic bacteria have significant detoxification potential in the body due to their metabolic activity and vast numbers in the body.
Probiotic bacteria can degrade harmful compounds, such as toxins and other carcinogenic compounds, into less harmful ones in the body.
This detoxification activity of probiotic bacteria is also targeted against infection-causing toxins released by pathogenic microorganisms that can establish themselves in the body.
It is also thought that the metabolic activity (see glossary) of probiotic bacteria explains the beneficial effect of probiotic supplementation for individuals with high cholesterol and digestive di culties.
What is the correct potency for probiotic products?
The biggest myth surrounding probiotic products is that the higher the potency of a probiotic product, the more effective that probiotic product will be for the customer. In fact, unlike other supplements, increasing the amount of live probiotic bacteria contained in a product does not necessarily increase the effectiveness of the product.
The key criteria for the potency of a probiotic product is that it contains a minimum threshold of live bacteria, around 2 billion (CFU 2×109), which can colonise and proliferate in the intestine. Increasing the number of bacteria above this minimum threshold does not increase the health benefit.
Factors such as stability, survivability (see glossary), inhibition of pathogens, and antibiotic resistance (all explained below) are ultimately more important to a probiotic product’s effectiveness, than the actual number of live probiotic bacteria contained in the product, provided that the product contains a minimum threshold of live bacteria, around 2 billion (CFU 2×109).
What determines the effectiveness of probiotic bacteria?
The potency of a probiotic product (sometimes referred to as “CFU”) is only a small factor in its effectiveness. More importantly, the probiotic bacteria strains used in the product must possess certain key characteristics, listed below, to be effective in conferring a health benefit.
Established probiotic brands like Quest invest in testing and detailing these characteristics in-house, to confirm the effectiveness and superiority of its probiotic products and probiotic bacteria strains.
Survivability
Probiotic bacteria must be able to survive the transit through the stomach so that they reach and colonise in the intestine to confer their health benefits.
Survivability characteristics of Quest probiotic strains are confirmed by testing their in vitro (see glossary) tolerance to the low (acidic) pH, bile salts and pancreatic medium, to which they will be exposed to during the transit through the stomach to the intestine.
Table 3: Acidic tolerance of Quest probiotic strains at pH3 (60 mins at 370C)
| Strain | Result | Acid tolerance (pH60 3mins 370C) |
| L. acidophilus | +++ | >75% survival |
| L. casei | ++ | >50% survival |
| L. rhamnosus | ++ | >50% survival |
| L. plantarum | +++ | >70% survival |
| L. bulgaricus | ++ | >50% survival |
| L. gasseri | ++ | >50% survival |
| L. paraplantarum | ++ | >50% survival |
| L. fermentum | ++ | >50% survival |
++++ Excellent +++ Very Good ++ Good + Fair
Table 4: Bile tolerance of Quest probiotic strains in 0.5% bile salt (26hrs at pH 5.0)
| Strain | Result | Bile Tolerance (26 %0.5hrs pH 5.0) |
| L. acidophilus | ++++ | >90% survival |
| L. casei | ++++ | >90% survival |
| L. rhamnosus | ++++ | >90% survival |
| L. plantarum | ++++ | >90% survival |
| L. bulgaricus | ++++ | >90% survival |
| L. gasseri | ++++ | >90% survival |
| L. paraplantarum | ++++ | >90% survival |
| L. fermentum | ++++ | >90% survival |
++++ Excellent +++ Very Good ++ Good + Fair
Table 5: Pancreatic medium tolerance of Quest probiotic strains in 0.2% pancreatic medium (180 mins at pH7.5)
| Strain | Result | Pancreatic Tolerance (180 %0.2min pH7.5) |
| L. acidophilus | +++ | >75% survival |
| L. casei | +++ | >75% survival |
| L. rhamnosus | +++ | >75% survival |
| L. plantarum | +++ | >75% survival |
| L. bulgaricus | +++ | >75% survival |
| L. gasseri | ++ | >50% survival |
| L. paraplantarum | +++ | >75% survival |
| L. fermentum | ++ | >50% survival |
++++ Excellent +++ Very Good ++ Good + Fair
Antibiotic resistance
Antibiotic resistance demonstrates the ability of a probiotic bacteria strain to survive and even grow in conditions that would normally inhibit or kill bacteria. As a result, antibiotic resistance is a key indicator to a probiotic strain’s effectiveness to colonise the intestine and confer health benefits.
Antibiotic resistance also supports the use of specific probiotic strains for the treatment of gastrointestinal side effects caused by the use of antibiotics, including antibiotic-induced diarrhoea.
Table 6: Antibiotic resistance of certain Quest probiotic strains
| Strain | Ka | Ox | Va | Me | Ge |
| L. acidophilus | R | S | S | R | R |
| L. casei | R | R | S | R | R |
| L. rhamnosus | R | R | R | R | I |
| L. plantarum | R | R | R | R | R |
| L. bulgaricus | R | R | R | R | I |
| L. gasseri | R | R | R | R | I |
| L. paraplantarum | R | I | R | R | I |
| L. fermentum | R | I | I | R | I |
R = Resistant I = Intermediate (some resistance) S=Sensitive
| KEY OF ANTIBIOTICS: | Ka = Kanamycin (30mcg) | Ox = Oxacillin (1mcg) |
| Va = Vancomycin (30mcg) | Me = Metronidazole (50mcg) | Ge = Gentamycin (10mcg) |
Inhibition of pathogens
The protective role of probiotic bacteria against pathogenic microorganisms is one of the key health bene ts of probiotics. However, the ability of probiotics to inhibit, displace and compete with such pathogens is strain-specific.
The ability of Quest probiotic strains to inhibit pathogenic microorganisms is confirmed by testing their inhibitory activity against Staphylococcus aureus, a bacteria that is linked to many human infections, including pneumonia, meningitis and MRSA.
Table 7: Antagonistic activity of certaiInnh Qibuiteiosnt Sp.Aruorbeuiostic strains to inhibit Staphylococcus aureus
| Strain | Result | Inhibition |
| L. acidophilus | ++++ | 20mm inhibition zone |
| L. casei | ++ | 10mm inhibition zone |
| L. rhamnosus | ++++ | 20mm inhibition zone |
| L. plantarum | ++++ | 22mm inhibition zone |
++++ Excellent +++ Very Good ++ Good + Fair
Adhesion to intestinal wall
A probiotic strain that can effectively and strongly bind to the cells on the intestinal wall can more effectively colonise the gastrointestinal system and exert a positive influence on the body’s immune system. Good adhesion to the intestinal wall also allows a probiotic bacteria to reinforce the intestinal lining and more effectively compete for adhesion sites with potentially pathogenic microganisms.
Lactic acid production
Lactic acid production is the end product of metabolism by lactobacilli probiotic bacteria (e.g. L.acidophilus). As a result, lactic acid production is one of the key indicators to correctly characterise a lactobacilli probiotic strain. Lactic acid can also inhibit pathogenic microorganisms and so helps probiotic bacteria to inhibit pathogens in the body. All Quest lactobacilli probiotic strains have been con rmed to produce lactic acid.
Why is probiotic stability important?
Should probiotics be refrigerated?
Only live probiotic bacteria can have a positive effect on health and well-being by colonising the gastrointestinal system and exerting a positive influence in the body. As a result, it is of critical importance that probiotic products are stable throughout their shelf life and contain live (but dormant) bacteria.
Refrigeration is one technique used to increase the stability of probiotic products, but for the highest quality probiotic products, refrigeration in not always necessary.
Quest uses a combination of advanced manufacturing techniques and detailed shelf-life stability testing to guarantee the stability of its probiotic products and guarantee that its products contain live bacteria when purchased by the consumer. Quest guarantee that its products are stable at room temperature and that refrigeration is only optional.
Are probiotic bacteria safe?
Lactobacilli probiotic bacteria have a long established safety for human consumption, which is supported by several published studies.29 Lactobacilli probiotic bacteria are also included on the Qualified Presumption of Safety List issued by the European Food Safety Authority.30
Clinically reported complications caused by probiotic supplementation are rare and mostly involve mild abdominal discomfort and flatulence. Generally, probiotic supplements are not recommended in the case of individuals with suppressed immune systems (e.g. individuals with HIV/AIDS, organ transplant patients, premature infants), due to a low risk of infection.
The first step in guaranteeing the safety of a specific probiotic bacteria strain is to establish its correct identity. Quest has conducted extensive laboratory and genetic characterisation tests to confirm and verify the identity of each of its proprietary probiotic bacteria strains used in its products.
Is probiotic supplementation recommended and safe for children?
Probiotic supplementation is highly recommended for infants and children. The intestinal microflora develops in the first few years of life. Once this development stage is over, the intestinal microflora is essentially set, playing an important factor in the health of the individual for the rest or his or her life. Probiotic supplementation helps in the healthy development of the intestinal microflora in infants and children.
Children are particularly susceptible to potentially harmful “bad” bacteria from food and the environment. Probiotics play a key role in protecting children, including from stomach bugs. Probiotic bacteria have also shown to be beneffcial for atopic dermatitis (eczema) and antibiotic-induced diarrhoea in children.
There is also significant emerging research on the benefits of probiotics for infant health. Research has shown probiotics can reduce infantile colic and may also increase infant weight and height growth in the first 6 months of life.
Probiotic supplementation is considered safe for children in good health. In 2004, the European Society of Pediatric Gastroenterology stated that: “probiotics so far used in clinical trials can be generally considered safe”, but urged for ongoing surveillance in high-risk children with pre-existing diseases.31
Why have probiotic health claims been rejected by the EU?
In 2011, the EFSA was tasked with the assessment of health claims for all nutrients contained in food supplements. Under pressure from lobbyists, the EFSA adopted a scientific policy that condemned thousands of well-established nutrient health claims, including for probiotics, to a negative assessment.
This EFSA scientific policy favours nutrient health claims designed for normal healthy individuals rather than health claims for specific disorders. Furthermore, the EFSA also places most importance on large-scale human intervention studies (within the budgetary scope of only the largest pharmaceutical and food companies rather than smaller human studies complemented with laboratory studies, which are accepted in countries like Japan. Finally, the EFSA have refused to accept that a positive affect on “intestinal microflora” is evidence to prove the impact of a probiotic bacteria on health.
The negative assessment of health claims for probiotics by the EFSA does not detract from the well-established and growing scientific and clinical evidence supporting the use of probiotic bacteria for key health benefits. See Table 1 and 2.
Ultimately, we expect that the consumers who derive health benefits from probiotic products on a daily basis will continue to
drive the research in supporting scientific data, in the medium to long term, until these health claims become approved and established.
How are probiotic bacteria manufactured?
Very few brands can claim that they manufacture their own probiotic bacteria from start to end, or that they have their own proprietary probiotic bacteria strains which belong to them. Manufacturing and developing probiotic products is a complex and expensive process requiring detailed consideration of issues such as strain origin, strain characteristics, fermentability,
probiotic drying techniques, fermentation yields and stability.
Quest uses the most advanced lyophilisation (freeze-drying) techniques to micro-encapsulate (see glossary) its probiotic bacteria and enhance their stability at room temperature. Lyophilisation (freeze-drying) is a more reliable and expensive technique than spray-drying and creates stable probiotic bacteria.
Figure 1: Steps involved in the development and manufacture of probiotic products
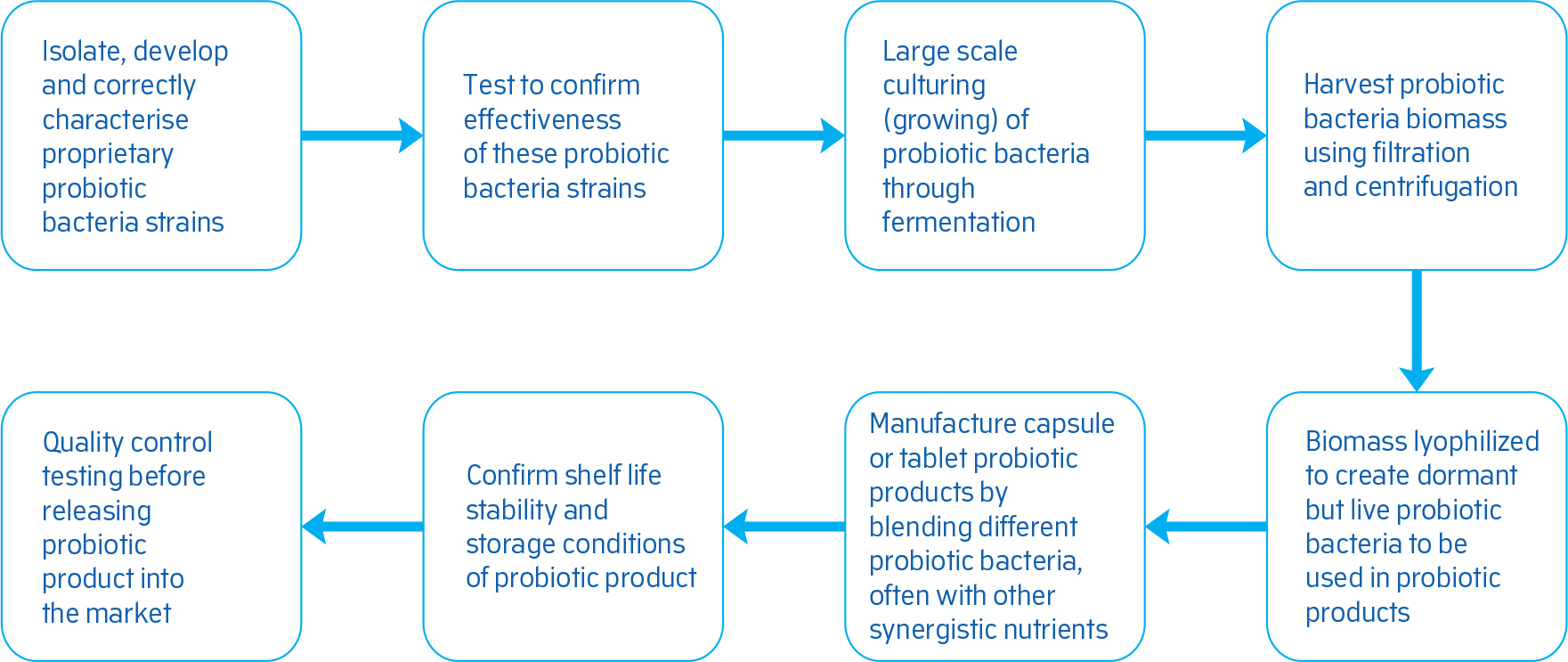
Figure 2: Images of Quest’s state-of-the-art probiotic manufacturing facility

Fermentation tanks culture (grow) probiotic biomass on a large-scale

Lyophilizer (freeze-dryer) creates dormant but live probiotic bacteria powder

Encapsulation of probiotic powder into 2-piece capsules
Glossary
Intestinal Micro ora (also referred to as Gut Flora) is the spectrum of bacteria, fungi and other microorganisms that live naturally within the intestine and form the living environment of the intestine.
Gastrointestinal System or Tract (also referred to as the Gut) refers to the stomach, intestine and large intestine.
Fermentation in the context of manufacturing probiotic bacteria, is the industrial growth of probiotic bacteria in fermentation tanks, by using nutrients, oxygen, agitation and warmth. The resulting “wet” biomass is then subjected to centrifugation and lyophilisation.
Inhibitory Compounds are chemical substances that inhibit the growth of microorganisms through various methods. These may be produced by microorganisms such as probiotic bacteria.
Microorganisms are microscopic living organisms, often single-celled, such as bacteria and fungi.
Pathogenic Microorganisms (also referred to as Pathogens) are microorganisms that produce disease or infection in their host. They can be bacterial, fungal, viral or parasites.
Lyophilisation (also referred to as Freeze-Drying) is the freeze-drying process that utilises very low temperatures and high pressure to remove water from probiotic bacteria creating a dry powder containing living but dormant probiotic bacteria, which can be incorporated into probiotic products such as tablets and capsules.
Metabolism (also referred to as Metabolic Activity) are the chemical reactions that occur inside the cells of living organisms and that sustain life. These reactions allow organisms to grow, reproduce, maintain their structures and respond to their environment. Metabolism will often use substances and compounds and will also create other useful substances and compounds as an end-product.
Survivability refers to the ability of a probiotic bacteria strain to survive the transit through the stomach, after being ingested, and to colonise inside the intestine.
In-Vitro refers to experiments relating to a living organism, but carried out in isolation outside the living-organism, usually in a laboratory.
Micro-encapsulate means microscopic encapsulation of a specific substance, in this case living probiotic bacteria, providing them with a protective outer-layer which can enhance their survivability.
Scientific References and Reviews
- Aliment Pharmacol Ther.1467-16:1461;2002
- Br Med J.1364-324:1361 ;2002
- Travel Med Infect Dis.105-5:97;2007
- Dig Dis Sci.2634-47:2625 ;2002
- Pediatrics.684-109:678;2002
- Cochrane Database Syst Rev 2.2004:CD003048
- BMC Gastroenterol.9:15;2009
- Gut.332-59:325;2010
- Am J Gastroenterol.103;2008(Suppl 1):S458
- FEMS Immunol Med Microbiol.134-35:131;2003
- Microecol Ther.45-23:32;1995
- Ann Intern Med.357-116:352;1992
- Swed Dent J.60-30:55;2005
- Int J Food Microbiol.223-95:219;2004
- Antimicrob Agents Chemother.1233-31:1231;1987
- Caries Res.420-35:412;2001
- J Allergy Clin Immunol.121-121:116;2008
- Ann Allergy Asthma Immunol.516-101:508;2010
- Am J Clin Dermatol.103-9:93;2008
- J Clin Microbiol.2330-27:2328;1989
- Aliment Pharmacol Ther.168-25:155;2007
- Ann Nutr Metab.27-54:22;2009
- Appl Environ Micobiol.1753-71:1745;2005
- World J Gastroenterol.75-16:69;2010
- Int J Antimicrob Agents.34:197;2009.e1-e10
- J Hum Nutr Diet.58-19:51;2006
- J Am Coll Nutr.419-25:415;2006
- Pediatrics.109;2007:e124-e130
- Int.J.Food Prot.106-44:93;1998
- http://www.efsa.europa.eu/EFSA/Scienti c_Opinion/sc_op_ej587_qps_en.pdf
- J Pediatr Gastroenterol Nutr.374-38:365;2004
- J Dairy Science.1606-78:1597;1995
- J Clin Gastroenterol.234-40:232;2006
- J Clin Gastroenterol.42;2008.Suppl96-2:591
- Gastroenterol Res Pract.2011:161358;2011
- Acta Diabetol.73-257:(4)48;2011
- PLoS One.(2)5;2010:e 9085
- Am J Clin Nutr.2000:(6)94;2011s-2005s
- Am J Clin Nutr. 2001 Feb;73(2 Suppl):421S-429S.
- Am J Clin Nutr. 2000 Dec;9-1474:(6)72.
Download Technical Paper
By entering your name and email address we may contact you by email or phone, subject to our privacy policy.

